Introduction
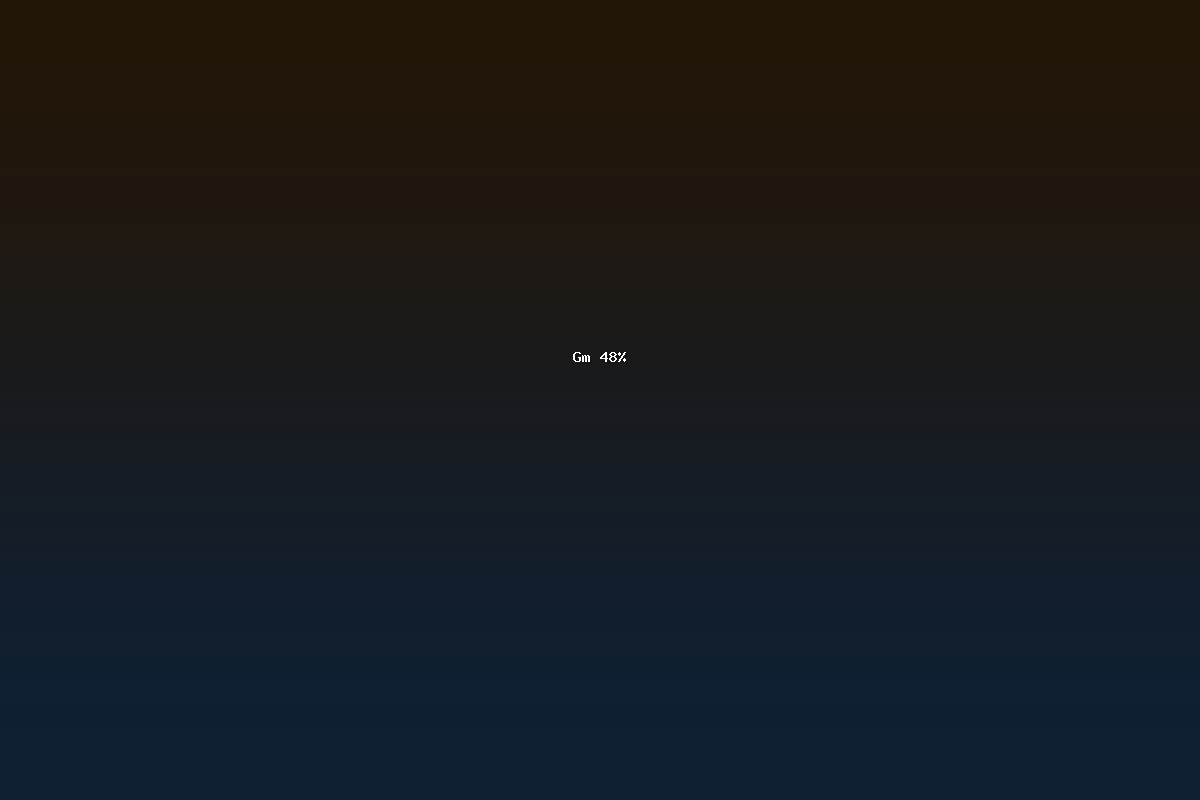
GM 48% is a quantitative descriptor that appears in the field of agricultural biotechnology and food science. The notation refers to a measured proportion, expressed as a percentage, of genetically modified (GM) material present in a sample, product, or process. In regulatory and scientific contexts, the term is used to report the level of genetically engineered content that meets or exceeds a specified threshold. The designation “48%” typically indicates that nearly half of the relevant biological or chemical material contains genetic modifications of a specific nature.
Although the term is concise, its usage is embedded in a complex framework of analytical techniques, regulatory policies, and industry practices. The value 48% may be the outcome of quantitative polymerase chain reaction (qPCR) assays, high‑throughput sequencing, or compositional analysis of biopharmaceuticals. Because the term is applied to diverse contexts - from crop seeds and animal feed to protein therapeutics - understanding GM 48% requires a multidisciplinary perspective that encompasses genetics, chemistry, law, and public policy.
This article provides an overview of GM 48% as a measurement concept, its historical development, technical foundations, regulatory significance, practical applications, and the debates that surround its use. The discussion draws on peer‑reviewed literature, international standards, and case studies from multiple jurisdictions.
History and Development
Early Use of Quantitative GM Indicators
Before the proliferation of GM organisms in the 1990s, assessments of genetic modification were largely qualitative. Researchers relied on Southern blotting and restriction fragment length polymorphism (RFLP) analyses to confirm the presence of inserted DNA sequences. As the number of commercialized GM crops increased, the need for quantitative measurements grew. In 1998, the International Organization for Standardization (ISO) published ISO 14430, a standard for the detection and quantification of GM DNA in food and feed.
The ISO 14430 protocol introduced qPCR as the primary technique for determining the percentage of GM content. The methodology involved comparing the cycle threshold (Ct) values of a target GM gene with an endogenous reference gene. This comparison yielded a relative quantity, which could be expressed as a percentage of GM material in the sample.
Adoption of the “% GM” Metric in Regulatory Frameworks
Regulatory agencies in the United States, European Union, Canada, and other regions adopted the “% GM” metric to set labeling thresholds. For instance, the U.S. Food and Drug Administration (FDA) established a 5% threshold for labeling GM ingredients in food products, whereas the European Union introduced a 0.9% threshold for certain crops. The designation “48%” has since been used in regulatory dossiers to indicate that a product exceeds these thresholds by a significant margin.
In the biopharmaceutical sector, the concept of GM 48% also emerged. Therapeutic proteins engineered in microbial or mammalian cell lines are often expressed at high levels; the percentage of product that contains the intended recombinant gene is quantified to ensure batch consistency and compliance with Good Manufacturing Practice (GMP) guidelines.
Standardization of Quantification Protocols
Over the past two decades, several consensus documents have refined the measurement of GM content. The Codex Alimentarius Commission’s “Guidelines for the Analysis of Genetically Modified Organisms” (1999) set the stage for harmonized methods. Subsequent updates in 2005 and 2014 incorporated next‑generation sequencing (NGS) and digital PCR (dPCR), improving sensitivity and specificity. These advancements enabled the reliable detection of low‑level GM material, including values such as 48%, which may otherwise fall near the detection limit of older assays.
In 2020, the International Federation of Pharmaceutical Manufacturers & Associations (IFPMA) published guidelines that required the quantification of recombinant DNA sequences in biologics. These guidelines specifically addressed the need to report percentages like 48% to document the proportion of product derived from a genetically engineered source.
Definition and Measurement
Conceptual Framework
GM 48% represents the relative proportion of a sample that carries a genetically engineered sequence relative to a reference sequence. In most contexts, the measurement is expressed as a mass or volume fraction (e.g., weight percent) rather than a simple count of molecules. The calculation typically follows these steps:
- Extraction of nucleic acid or protein from the sample.
- Amplification of target GM sequences using qPCR or dPCR.
- Amplification of endogenous reference sequences.
- Comparison of amplification curves to determine the relative abundance of GM versus reference material.
- Conversion of the relative abundance to a percentage, often using standard curves derived from known mixtures.
While the principle is straightforward, the accuracy of the percentage depends on the quality of reagents, the efficiency of amplification, and the homogeneity of the sample. Laboratories typically validate their methods using control samples that contain known percentages of GM content, including the 48% level.
Analytical Techniques
Quantitative Polymerase Chain Reaction (qPCR)
qPCR remains the most widely used method for measuring GM content in food and feed. The technique employs fluorescent probes that bind to the target sequence, allowing real‑time monitoring of amplification. By comparing Ct values for the GM target and a reference gene, the method can estimate the relative quantity. Calibration curves built from mixtures of GM and non‑GM material facilitate conversion to absolute percentages.
Digital PCR (dPCR)
dPCR partitions the sample into thousands of nanoliter reactions, each containing zero or one target molecule. After amplification, the number of positive partitions is counted, yielding an absolute quantification of the target sequence. dPCR offers higher precision, particularly for low‑level detection, and is therefore used for regulatory compliance when values near threshold limits are involved.
Next‑Generation Sequencing (NGS)
NGS technologies, such as Illumina sequencing, provide comprehensive coverage of the sample genome. By mapping reads to reference genomes and counting reads that map to engineered sequences, laboratories can determine the proportion of GM material. Although more resource‑intensive, NGS offers the advantage of detecting unexpected genetic modifications.
Protein‑Based Assays
In the biopharmaceutical domain, enzyme‑linked immunosorbent assays (ELISA) or mass spectrometry are employed to quantify recombinant proteins. The relative abundance of the recombinant protein compared to total protein content can be expressed as a percentage, analogous to GM 48% in nucleic acid assays.
Quality Control and Validation
Laboratory protocols for measuring GM 48% must adhere to ISO 17025 and, where applicable, Good Laboratory Practice (GLP). Validation parameters include:
- Accuracy: Comparison with certified reference materials.
- Precision: Repeatability and reproducibility studies.
- Specificity: Ability to discriminate the target GM sequence from closely related wild‑type sequences.
- Limit of Detection (LOD) and Limit of Quantification (LOQ): Minimum detectable and quantifiable percentages.
Validation ensures that reported percentages, such as 48%, reflect true composition rather than analytical artifacts.
Regulatory Frameworks
International Standards
Several international bodies have codified guidelines for GM content measurement:
- ISO 14430 (Food and feed: Detection and quantification of genetically modified organisms by polymerase chain reaction).
- Codex Alimentarius: Guidelines for the Analysis of GMOs.
- International Organization for Standardization (ISO) 21594: Quantitative methods for detecting genetically modified DNA.
- IFPMA Guidelines for Recombinant DNA Quantification in Biologics.
These standards set technical requirements that laboratories must meet to produce reliable GM 48% measurements. Compliance is often verified through interlaboratory comparisons and proficiency testing.
Regulatory Thresholds by Region
Regulatory thresholds define the minimum GM content that triggers labeling, import restrictions, or mandatory testing. While thresholds differ across regions, the 48% level frequently exceeds these limits, indicating significant GM presence.
United States
In the U.S., the FDA requires labeling of foods containing 5% or more of GM ingredients. The U.S. Department of Agriculture (USDA) requires labeling for crops engineered for insect resistance and herbicide tolerance. The National Organic Program also prohibits GM materials in organic production.
European Union
The EU’s Regulation (EC) No 1829/2003 mandates labeling for foods containing GMOs at levels above 0.9%. The European Food Safety Authority (EFSA) oversees risk assessments and sets thresholds for specific GM crops, such as maize and soybean.
Canada
Canada’s Health Canada and Agriculture and Agri‑Food Canada impose a 1% labeling threshold for GM foods. The country also requires import testing for GM content above certain levels.
Australia and New Zealand
Both countries have a 0.1% labeling threshold for GM foods. Australia’s Department of Agriculture, Water and the Environment monitors import consignments for GM content.
South Africa and Argentina
South Africa’s legislation requires labeling for GM foods at 1% or higher. Argentina’s regulations also impose labeling thresholds, with a 0.5% minimum for certain crops.
Implications of GM 48% for Compliance
When a product’s GM content is quantified as 48%, it typically falls well above regulatory thresholds. Consequently, the product must carry a GM label, be excluded from organic certification, and may face restrictions in markets with lower thresholds. Importers must provide documentation of GM content, and failure to comply can result in product rejection, fines, or other penalties.
Applications in Agriculture
Seed Production and Distribution
Seed companies often report GM percentages in their product specifications to assure growers of purity. A 48% GM level indicates a high proportion of engineered seed, which may be desirable for certain pest‑resistant varieties. Seed blends with mixed GM content are also labeled with percentages to inform agronomists and farmers.
Animal Feed
Animal feed manufacturers incorporate GM grains, such as soybean meal and corn, into feed formulations. Reporting GM 48% ensures transparency and compliance with labeling mandates for animal products destined for human consumption. For example, meat, dairy, and egg producers may request or require that their feed contain less than a specified GM threshold to meet market demands for non‑GM or organic products.
Food Processing and Labeling
Food processors must test raw ingredients to verify GM content. When the measured GM percentage is 48%, processors must label the final product accordingly. This is particularly relevant for processed foods containing soy, corn, or canola oil. In the U.S., the FDA’s 5% labeling threshold means that a product with 48% GM content would carry a mandatory “Contains GMOs” statement.
Biopharmaceutical Production
Biologics, such as monoclonal antibodies and vaccines, are produced in genetically engineered cell lines. The 48% figure may represent the proportion of the protein batch that contains the recombinant gene product. Ensuring a high GM percentage is critical for maintaining batch consistency, potency, and safety. Regulatory agencies require that manufacturers provide quantitative data, including percentages like 48%, to demonstrate compliance with GMP.
Industrial Biotechnology
Enzymes, biofuels, and specialty chemicals derived from GM microorganisms often have production yields quantified in terms of GM content. A 48% value indicates the proportion of product derived from engineered cells, which can impact cost, downstream purification, and environmental assessment.
Research and Development
Academic laboratories use GM 48% as a benchmark for evaluating transformation efficiency. For example, when transfecting plant cells, researchers may report that 48% of the resulting cell lines carry the transgene, providing a measure of success for the protocol. Similarly, in gene editing experiments, a 48% editing rate indicates that nearly half of the cells underwent the intended modification.
Environmental and Health Impacts
Ecological Consequences
High levels of GM content in crops can influence biodiversity, gene flow, and ecosystem dynamics. A product with 48% GM content, such as a field trial of engineered corn, may release transgenes into wild relatives or non‑target organisms. Studies have examined gene flow from GM maize to wild relatives, with findings indicating variable rates depending on the crop, geography, and environmental conditions. The potential for horizontal gene transfer remains a subject of ongoing research.
Human Health Considerations
Regulatory agencies assess GM products for allergenicity, toxicity, and nutritional equivalence. The presence of 48% GM material in a food product requires a thorough safety assessment to ensure that engineered components do not pose risks to consumers. In many cases, the safety profile of GM crops, such as Bt maize, has been established through extensive testing, leading to regulatory approval in multiple regions. However, consumer perception often diverges from scientific consensus, with some segments of the population expressing concerns over GM content.
Food Quality and Nutrition
Some GM crops have been engineered to enhance nutritional content, such as vitamin‑A‑rich “Golden Rice.” The 48% GM level ensures a significant amount of the target trait, but the overall nutrient profile of the food must still meet regulatory and consumer expectations. Nutrient labeling may include specific statements about the GM content’s contribution to nutritional content.
Socioeconomic Impacts
The adoption of GM crops with high GM percentages, like 48%, can affect trade flows, market access, and agricultural practices. Producers may gain cost advantages from reduced pesticide use or increased yield but may also face restrictions in markets with lower GM thresholds. The socioeconomic balance between innovation benefits and consumer preferences is a key aspect of policy discussions surrounding GM content reporting.
Consumer Perspectives
Transparency and Trust
Consumers increasingly demand transparency regarding GM content. The 48% figure, when disclosed, informs purchasing decisions and can influence brand perception. Some consumers reject products with GM content above their personal thresholds, while others accept GM foods if the product offers clear benefits, such as lower pesticide use or environmental sustainability.
Market Segmentation
Retailers differentiate products based on GM content. Non‑GM or organic categories attract consumers willing to pay premium prices. Products with 48% GM content may be positioned as “All‑organic” or “Certified Non‑GM” only after dilution or processing to reduce GM content below thresholds. The labeling of GM percentages can also serve as a marketing tool for niche markets, such as “Reduced‑Pesticide” labeling.
Public Perception
Public sentiment towards GM products varies by region, age group, and cultural context. In the U.S., a 48% GM product is commonly labeled and often accepted by the majority of consumers. In contrast, European markets exhibit stronger skepticism toward GM content, leading to stricter labeling and higher rejection rates. Understanding consumer attitudes helps companies tailor product strategies and communication efforts.
Future Trends
Advancements in Gene Editing
CRISPR‑Cas9 and related technologies are pushing transformation efficiencies beyond traditional methods. As editing precision improves, reporting editing rates in percentages, such as 48%, becomes more common. This trend may enable higher transformation efficiencies, potentially exceeding 80% or more, thereby redefining benchmarks like 48%.
Metabolic Engineering
Engineering metabolic pathways in crops and microbes can increase the GM percentage of produced metabolites. For example, yeast engineered to produce high‑value oils may achieve 48% GM content in the final extraction, enhancing yield while simplifying downstream processing.
Regulatory Harmonization
There is an ongoing push toward harmonizing GM content standards worldwide. The International Union of Food Science and Technology (IUFOS) and other organizations aim to align testing protocols and thresholds. A harmonized framework would streamline cross‑border trade and reduce the complexity of reporting GM 48% data in multiple formats.
Data Transparency and Public Databases
Open access databases, such as the USDA’s National Genetic Engineering Database (NGEDB) and EFSA’s GMO Database, store GM content data. Reporting percentages like 48% in these databases facilitates public scrutiny, academic research, and policy development. Data transparency also supports consumer education and trust building.
Conclusion
GM 48% is a quantitative measure that informs a wide range of stakeholders - regulators, producers, consumers, and researchers - about the extent of genetic modification in a product. Its measurement relies on validated analytical methods, and its implications for compliance, labeling, and market access are significant. In agriculture, a 48% GM level typically indicates a substantial presence of engineered material, affecting seed, feed, food, and biopharmaceutical production. While regulatory frameworks largely treat values above 5% (U.S.) or 0.9% (EU) as requiring labeling, a 48% figure ensures that products are clearly identified as containing GMOs. Consequently, transparency and accurate reporting of GM percentages are essential for maintaining consumer trust, ensuring food safety, and navigating international trade rules.





No comments yet. Be the first to comment!